E10 compatibility issues
Material compatibility and safety issues are handled extensively in the Guidebooks, e.g. Engelen et al. (2008), RFA (2010) and E85 Handbook (2008).
How much ethanol can be used in conventional cars?
Ethanol/gasoline blends have been used for decades in some countries, for example “gasohol” has been used since 70’s in the U.S. and Brazil. The U.S. Environmental Protection Agency (EPA) accepts gasoline that contains 10-15 vol-% ethanol for 2001 and newer light-duty vehicles, subject to certain conditions (US EPA 2011). In Brazil, ethanol content of gasohol has gradually risen to 25 vol-% for use in slightly modified cars. In Europe, maximum limit for ethanol content in gasoline is 10 vol-% (E10) since 2011, but maximum 5 vol-% ethanol containing gasoline is available for cars, which are not compatible with E10 (Directive 2009/30/EC).
Mid-level ethanol blending ratios are under research in many regions. Some results have shown that gasoline engines from 2001 through 2009 may fail when operated on E15 and E20 (CRC 2012). Many sites also require modifications into dispensing system for selling E15 (Gregory 2012). On the other hand, some results show that E15 has no adverse effect in the 1994-2004 model year U.S. light-duty vehicles (Ricardo 2010).
Compatibility of ethanol with conventional cars depends on technology and material choices. Modern cars with a closed-loop fuel control system can compensate the leaning effect of oxygen containing gasoline and modern gasoline fuelled cars are often built up with materials that tolerate ethanol. However, fuel injection system of other than FFVs for E85 use is not compatible with high concentration ethanol. Therefore maximum oxygen content for gasoline is defined in legislation and standards. (Read more). Renewal of car population is a slow process. JEC (2011) has estimated that in 2020 about 10% of European car fleet is older than model year 2005. Older cars and non-road engines are less compatible with ethanol than modern cars.
Driveability is affected by volatility of fuel. Volatility is specified by vapor pressure and distillation properties of gasoline (Read more). These properties are controlled in legislation and standards to ensure proper cold-starting, warm-up behavior, driveability, and to avoid unnecessarily high evaporative emissions. Modern cars with multi-point fuel injection systems are generally less sensitive towards volatility properties of gasoline than old cars. With old cars, front-part of distillation was critical for cold-start performance and evaporative emissions, whereas mid-range distillation was important for warm-up performance, acceleration and short-trip economy. End-part of distillation influenced oil dilution, deposits and long-trip economy.
When ethanol is added to gasoline, it influences the front part of distillation (Read more) and may affect hot-weather drivability. Vapor in this part of distillation contains an even higher fraction of ethanol than would be expected based on its share in the gasoline, due to the azeotropic behavior of the ethanol/gasoline blend. Modern vehicles using multi-point injection technology are less susceptible to hot-weather driveability problems than older vehicles. The EN228 specification appears adequate for controlling drivability in European vehicles, but further studies are needed if the standard’s volatility limits are modified to allow ethanol blends of 10 vol-% and higher. (Stradling et al. 2009).
Cold-weather drivability is affected by mid-range volatility, defined in Europe by the E100 value. This parameter is linked to exhaust emissions under cold-start conditions. With splash blends of ethanol in gasoline, cold-weather drivability may improve somewhat due to the higher volatility of the blends, but the opposite was observed when fuels were tailored at the same volatility levels. Ethanol’s high latent heat may affect drivability with a cold engine, and may also have a leaning effect under open-loop engine conditions. Stradling et al. (2009) suggest that minimum E100 limits in the EN228 gasoline specification would ideally vary with the ambient temperature at different volatility classes. The CRC in the USA has developed new fuel parameters (Driveability Indices) that include ethanol offset terms for US vehicles. Ethanol would prevent icing problems, which are, however, probably relevant only for some old vehicles.
Ethanol is more aggressive towards materials than gasoline. This may lead to early wear or damage to materials and components, swelling of elastomers and corrosion of metals. Ethanol may dissolve lubricant layers between metal parts, which may increase wear. The high electrical conductivity of ethanol may lead to a risk of electrolytic or galvanic corrosion with materials designed for hydrocarbons (Walwijk et al. 1996). Corrosion inhibitors are recommended for ethanol, and compatibility of additives should be secured (RFA 2010, Engelen et al. 2008).
In Brazil, there is long experience of blends of around 20% ethanol with gasoline. Examples of modifications to cars relating to this fuel include cylinder walls, cylinder heads, valves, valve seats, pistons, piston rings, intake manifolds, carburetors, electrical systems and nickel plating on steel lines.
Consideration must be given to alcohol/gasoline blends in the distribution chain. Materials used in tankers, tanks, lines, seals, hoses, and refueling equipment must be alcohol-resistant.
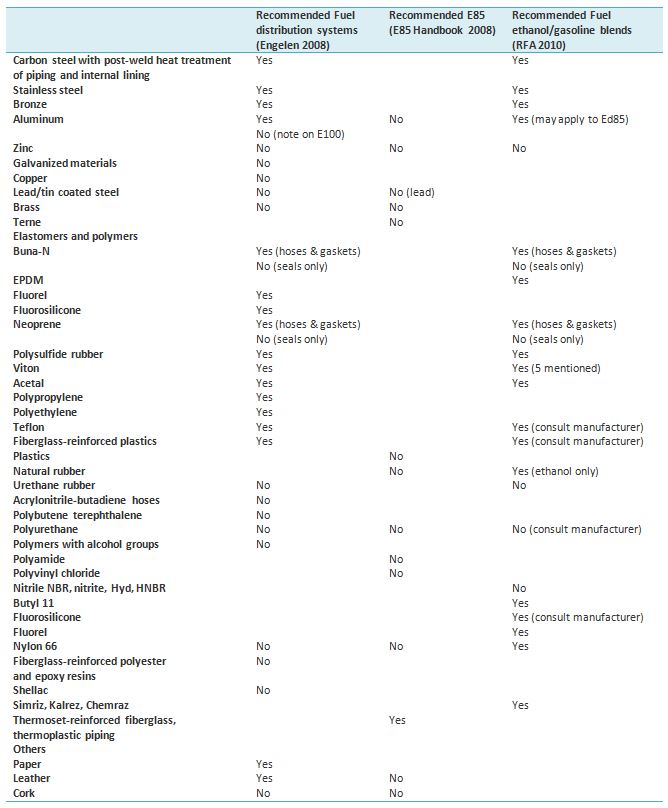
Material recommendations for ethanol and ethanol/gasoline blends are listed in Table 1. Recommendations are listed by Engelen (2008) and RFA (2010). The material cautions defined for the E85 fuel are included, as well (E85 Handbook 2013). Recommendations focus on materials of vehicles and/or fuel distribution systems. In particular, aluminum should be avoided. First-generation direct-injection fuel systems with aluminum rails do not tolerate ethanol (ETP 2011). Corrosion product of aluminum forms a gel. Water may suppress the reaction between alcohol and aluminum (Kameoka et al. 2005). Engelen et al. (2008) point out that manufacturers of products should be consulted for before introducing ethanol or ethanol/gasoline blends. Ethanol may also damage conventional paints, which is a considerable risk during refueling.
E25 dispensers may not be compatible with E85 (E85 Handbook 2013). For example, blender pump dipenser for E85 should have filters with nominal rating of 50% for 5 microns and absolute rating of 99% for 10 micron particles, whereas 10-micron filter is typically used for gasoline and low-level ethanol blends, and 30-micron filter for diesel fuel.
Table 1. Recommendations for materials considered for use in ethanol and ethanol/gasoline blends in fuel distribution system by Engelen et al. (2008) and recommendations for E85 (E85 Handbook 2008).
The storage and stability of ethanol blends are special issues due to ethanol’s affinity for water and the risk of phase separation. In addition, ethanol tends to clean impurities from fuel tanks and lines leading to risk of plugging the fuel filter.
The effect of water differs significantly depending on whether the water is dissolved in gasoline or is in a separate phase. A small amount of water in a homogeneous fuel has no adverse effect. If phase separation occurs, the ethanol/water mixture stays as a layer at the bottom of the tank and the octane number of the gasoline layer falls. The engine stalls if it takes in the ethanol/water phase. Furthermore, a mixture of ethanol and water is corrosive.
The risk of phase separation depends on the temperature and the aromatics and ethanol contents of gasoline. At low temperatures, the risk of phase separation increases. The higher the ethanol and aromatic contents of gasoline, the higher the proportion of water that can be absorbed by the fuel without phase separation. Phase separation may occur when the critical conditions change. For example, if a tank containing a little ethanol blend is filled with gasoline, the concentration of ethanol in the blend falls. In this situation, the presence of the water originally dissolved in the fuel may be sufficient to precipitate phase separation. The same may happen if the ambient temperature falls. The risk of phase separation is lower if stabilizing agents are used.
To avoid phase separation, anhydrous ethanol is used for low-level ethanol/gasoline blends. The whole chain from supplier to dispenser must be water-free. In practice, anhydrous ethanol is transported by tankers and stored in tanks specially designed for it. Low-level ethanol blends are typically blended at a product terminal or dispenser just before delivery to retail or the end user. Additional cleaning procedures may be needed for ethanol pipelines and tanks, depending on the market area (Owen and Coley 1995). E85 Handbook (2013) lists cleaning procedures, such as optic sweep, steam cleaning, filter agitator and chemical solvents.In the Netherlands, 15% hydrous ethanol (HE15) has been studied as fuel for gasoline cars (http://www.heblends.com/).
When non-oxygenated gasoline is used, water accumulates at the bottom of the tank. With alcohol-containing gasoline, alcohol content of this water layer increases, and gradually the tank "dries". Alcohol is sometimes added to fuel for this purpose. If there is a lot of water at the bottom of the tank, there is a risk of the alcohol/water layer being drawn into the engine.
An example of water tolerance of ethanol/gasoline blends is shown if the Figure 1 (link to Ternary phase diagrams).
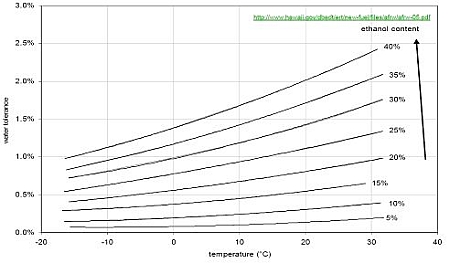
Figure 1. An example of water tolerance of ethanol/gasoline blends at different temperatures (Filho 2008).
With neat ethanol, the vapor in the air space in the fuel tank is flammable over a wide temperature range of about 12-43 °C (Engelen et al. 2008). With gasoline the mixture is too rich, and with diesel too lean, to be ignitable in the normal ambient temperature range. For neat ethanol, the flammability limits are wide. Vapor barriers or vapor return systems may be needed in the refueling system to avoid the risk of explosion and poisoning (McCormick 2001). In a study by NREL headspace vapors of gasoline samples became flammable at temperatures below -19 °C. Ethanol and gasoline vapors are denser than air and tend to settle in low areas (E85 Handbook 2013). However, the flammability risk with E10 fuel is evaluated to be close to that of gasoline (Paasi et al. 2008).
Alcohol resistant foams are needed in fires with high concentration ethanol, whereas fire-fighting chemicals traditionally used for gasoline are insufficient. Neat ethanol burns without smoke or visible flames, whereas flame of fuel ethanol is visible in daylight. (Engelen et al. 2008, E85 Handbook 2013).
High electrical conductivity of fuel in general increases safety by dissipating static charge e.g. in refueling. However, capability of a substance to generate and accumulate electrostatic charge is a complex phenomenon. For example, impurities, immiscibles, and multiple phases can exacerbate electrostatic charges. (Stephenson 2004).
Denaturants are required in fuel ethanol to make it toxic. Denaturants can impact engine operation and thus only accepted denaturants should be used (Legislation and standards).
If fuel tanks leak, ethanol and gasoline get into the ground. If groundwater is present, ethanol dissolves in it and is biodegraded. One factor that has been studied is the effect of ethanol on the transportation of gasoline in the ground and in groundwater. Deep et al. (2002) reported the effect of ethanol on the rate of benzene biodegradation under non-limiting oxygen and nutrient conditions. The biodegradation of benzene was severely inhibited in the presence of ethanol. Ethanol is degraded preferentially to benzene, which means that ethanol has an impact on benzene plume lengths in subsurface environments. Model simulations indicated that benzene plume lengths are likely to increase by 16–34% in the presence of ethanol.
Lahvis (2003) reported the potential effects of low-volume releases of ethanol-blended gasoline in the vadose zone. Model results indicate that the migration of ethanol in the vadose zone is limited to less than 100 cm from the source for releases in sand. In sandy clay, ethanol transport is limited to less than 50 cm. Furthermore, the presence of ethanol in gasoline does not significantly affect benzene transport and mass loading to groundwater. Travel times to groundwater may be more than an order of magnitude greater for ethanol than for benzene, depending on conditions. The model results indicate that the impacts of ethanol and benzene on groundwater from low-volume releases of ethanol-blended gasoline in the vadose zone are not expected to be significant unless the release is near the water table (< 100 cm) or, in the case of benzene, its biodegradation is limited.
Only a limited number of studies on the exhaust emissions with ethanol have been published with cars from 2000s. Studies with cars from 90’s are relevant to some extent, whereas older studies with carbureted cars do not represent today’s car fleet. Major part of studies have been carried out with 10% ethanol containing gasoline, but tests on mid-level ethanol blends have also been reported, e.g. by Hammel-Smith (2002) and Karlsson (2006).
A number of reviews on ethanol studies have been conducted. One of the reviews carried out within IEA Advanced Motor Fuels program was published by Larsen et al. (2009). Karman (2003) reviewed studies, which were published after 1997. Niven (2005) reviewed studies from 90’s and from early 2000’s. These studies are summarized by AFDC (2009).
Evaporative emissions are accumulated by permeation, leaks (liquid and vapor) and fuel tank venting (canister losses). Refueling produces volatile organic emissions (Larsen et al. 2009, Haskew et al. 2006).
Ethanol may increase the permeation rate through fuel system. Ethanol is a polar compound and a smaller molecule than typical hydrocarbons, and can pass walls of fuel system. (Reynolds 2002). The influence of ethanol on permeation seems to be highest at the low ethanol concentration (Figure 2). (Stahl et al. 1992 in Kassel 2006).
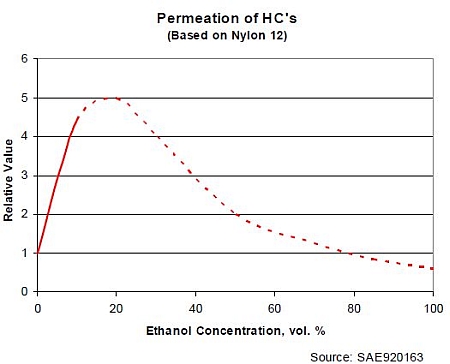
Figure 2. The effect of ethanol on permeation. (Stahl et al. 1992 in Kassel 2006).
Generally, low-level ethanol/gasoline blends tend to increase evaporative emissions. However, the results from different studies are not consistent. With “splash blended”, the vapor pressure is higher for low concentration ethanol blend than for gasoline. With “tailored” fuels, vapor pressure of ethanol blends are adjusted by using low volatility baseline gasoline (Vapor pressure). In some areas, legislation allows higher vapor pressure limit for ethanol containing gasoline.
Variation between the evaporative emission results with different cars is thought to be due to factors such as design and volume of carbon canister and vapor system. The cars with small carbon canisters compared to tank volume struggle with capacity, whereas larger canisters cope better with increased vapor pressure. (Australia 2008). Martini (2007) reported that a reduction in canister working capacity due to ethanol may increase evaporative emission. Studies in 70’s and 80’s addressed higher binding efficiency of ethanol to the activated carbons, as well as tendency of hygroscopic ethanol to carry water in carbon canister (Croes et al. 1999). Martini (2007) reported that heavy hydrocarbons and ethanol are hard to purge from active carbon in the canister. A trace-effect of ethanol may be seen after the tests with ethanol-containing fuels. Some carbon types preferentially absorb ethanol whilst others do not. Even 8 years old vehicles may emit artifacts, remnants of the solvents and adhesives, which is challenging for experimental work. (Australian study 2008).
Examples of studies, which do not show an increase in evaporative emissions with low-level ethanol blend
- Graham et al. (2008) did not find any statistically significant differences in diurnal and hot soak losses between E0, E10, E20 or splash-blended E10 fuel. Ethanol concentration of evaporative emissions followed the ethanol concentration of fuel. The MPFI and GDI cars, model years from 1998 to 2003 were tested +20 and -10 °C.
- Martini (2007) and Concawe (2006) reported that evaporative emissions from cars were dependent on vapor pressure of fuel, not on the ethanol content of fuel. Ethanol blends with vapour pressure around 75 kPa showed higher evaporative emissions than the fuels with vapour pressure in the range of 60-70 kPa, whether or not they contained ethanol. Conditioning carbon canisters of cars between the tests was challenging. Martini (2007) observed that evaporative emissions contained relatively high levels of light hydrocarbons (C4-C6), low levels of ethanol and significant concentrations of heavier hydrocarbons, like aromatics. The main sources of the light hydrocarbons are canister bleed emissions and breathing losses, while heavier hydrocarbons may originate through fuel permeation.
Examples of studies, which show an increase in evaporative emissions with low-level ethanol blends
- A 20-80% increase in evaporative emissions was observed with splash-blended E10 (AFDC 2009)
- At least double hot soak and diurnal losses with splash-blended E5 when compared to baseline gasoline. Benzene and aromatic emissions followed similar patterns as total evaporative hydrocarbon emissions. Alcohol emissions followed alcohol content of fuel, but not with all cars. (Australian study 2008)
- Tailored low-level ethanol blends did not increase refueling loss emissions, whereas running loss emissions with ethanol increased more than with gasoline. This was thought to be due to faster increase in vapor pressure with increasing temperature for ethanol than that for gasoline (link). In another referred study ethanol increased diurnal breathing losses likely due to differences in permeation abilities of fuels. (Wallace et al. 2009 references).
- Ethanol blends increased diurnal permeation rates compared to E0 gasoline, but no significant differences in ethanol or aromatics contents was observed (Haskew et al. 2006). Despite of higher evaporative emissions with ethanol blends, the average specific reactivities of the permeates were lower for the ethanol blends than for E0. The diurnal permeation rate was on average from 177 mg/day for E0, 484 mg/day for E10 and only 36 to 64 mg/day for “Zero Fuel Evaporative Emission” car. Study included E0, E6, E10 and E20 fuels and model year 2000, 2001 and 2004 vehicles.
- In the in-use program in Sweden 20 of 50 cars tested exceeded the 2 grams limit of evaporative emissions, which was likely due to 5% ethanol content of gasoline in Sweden. (Åsman 2006).
- The level of evaporated HCs was around 200 ppm with 63 kPa gasoline, and 340 ppm with E10 blended in the 70 kPa gasoline. Butane was used to adjust vapor pressure of gasoline, and it was the main component that vaporized during the tests. The evaporation of ethanol followed the similar trend as evaporated HCs. (Egebäck 2005).
- Tailored E10 decreased diurnal, but increased hot soak evaporative emissions ending up to a slight increase of evaporative emissions. Higher hot soak emissions were explained by distillation characteristics. (Environment Australia 2002 references).
Larsen et al. (2009) reviewed literature on the exhaust emissions with ethanol blends, and noticed that results regarding NOx emissions are contradictory. In the old studied, differences in NOx emissions with low-level ethanol blends ranged from 5% decrease to 5% increase when compared to base-line gasoline (CRFA 2003). A study with six cars from 90's showed an increase in NOx emission at higher ethanol concentrations than 12 vol-% (Environment Australia 2002). An Australian study (2008) did not find a clear trend for the NOx emissions.
Graham et al. (2008) observed that NOx emission increased as ethanol content increased mainly during the cold-start of test cycle and during aggressive driving conditions. There was no significant difference in NOx emissions during stabilized driving. Increase in NOx emission was substantial for E20 fuel at -20 °C.
Durbin et al. (2006) observed that the effect of ethanol content on NOx emissions may be dependent on the distillation characteristics. NOx emissions increased with increasing ethanol content at the low T50 (50 vol-% evaporated temperature), but no effect was seen at the higher T50 level.
The behaviour of the NOx emission with ethanol-containing fuels was car-dependent in a study by Aakko-Saksa et al. (2011). NOx emission decreased with increasing ethanol content for the FFV, whereas it increased for the MPFI and direct-injection gasoline cars at -7 °C.
Flame temperatures for alcohols are lower than for e.g., aromatics, which could lead to lower NOx emissions for alcohol fuels than for gasoline. However, this is not the case in old cars without close-loop control due to the leaning effect that raises combustion temperature. With modern cars, an increase in NOx emissions with ethanol may indicate lean mixture in some driving conditions with an influence on the performance of the exhaust catalyst.
Graham et al. (2008) reported that the nitrous oxide emission (N2O), strong greenhouse gas, tends to increase with increasing ethanol content. Formation of N2O depends on the performance of the exhaust catalyst, which may generate N2O at certain driving conditions. Old cars from 70’s and 80’s did not show significant influence of 10% ethanol blend on N2O emission (Environment Australia 2002).
Carbon monoxide and hydrocarbons
Larsen et al. (2009) reviewed literature on the exhaust emissions with ethanol blends. They reported that tailpipe CO and HC emissions generally reduced with increasing ethanol content. This was a conclusion also in the reviews by AFDC (2009), Niven (2005) and Karman (2003).
With old cars, enleanment of air to fuel ratio with ethanol leads to a decrease in CO and HC emission compared to baseline gasoline. For instance, in one study E10 fuel resulted in a 25-30% reduction in CO emission, and about 7% reduction in HC emission when compared to baseline gasoline. (CRFA 2003).
Australian study (2008) reported that CO and HC emissions decreased with E5 and E10 fuels compared to baseline gasoline, but the trends for new cars varied more and absolute differences were smaller than for older cars. For cars with closed-loop systems and catalysts, benefits in CO and HC emissions with ethanol are gained mainly at cold start or heavy acceleration (Graham 2008, Environment Australia 2002).
Graham et al. (2008) reported that tailored E10 showed lower CO emissions than baseline gasoline at normal and at -10 °C temperatures, whereas splash-blended E10 resulted in 35-50% higher CO emissions than tailored E10 at cold test temperature. At 20 °C, there was no statistical difference in NMHC or NMOG emissions between the fuels.
Durbin et al (2006) studied the effect of ethanol (0, 5.7, and 10 vol-%) and volatility parameters (mid-range and back-end volatility) on the exhaust emissions. CO emission decreased by 6-18% when ethanol content of fuel increased from 0 to 10%, depending on T50 temperature. NMHC emissions increased with increasing ethanol content, if distillation T90 temperature was high.
1,3-Butadiene, benzene and methane
Generally, tailpipe benzene and 1,3-butadiene emissions reduce with increasing ethanol content (Larsen et al. 2009, Niven 2005 and Karman 2003, Graham 2008, Aakko-Saksa et al. 2011). If ethanol is splash-blended gasoline, dilution effect leads to lower content of aromatics and olefins in gasoline resulting in lower benzene and 1,3-butadiene emissions. For non-consistent results on emissions of benzene and 1,3-butadiene emissions with ethanol blended fuels has been thought to be due to the performance of the exhaust catalyst in the cold-start (Environment Australia 2008). Durbin et al. (2006) noticed that in addition to ethanol content also distillation characteristics have an impact on the benzene and 1,3-butadiene emissions.
Larsen et al. (2009) reported that methane emissions increase when ethanol is added into gasoline, whereas Graham et al. (2008) reported that methane emissions seemed to decrease with increasing ethanol content of fuel.
Acetaldehyde emission increases substantially when ethanol containing fuels are compared to gasoline. Formaldehyde emission increases also when ethanol is used as low level blending component for gasoline. (Environment Australia 2008, Graham 2008, Durbin 2006, Aakko-Saksa et al. 2011).
In the study by Durbin et al. (2006), addition of 10% ethanol into gasoline increased acetaldehyde emissions by 73%. In this study, an interaction between ethanol content and the T50 temperature was observed. Graham et al. (2008) observed increase in acetaldehyde with both splash-blended and tailored E10.
An increase in acetaldehyde emissions have been observed mainly during cold-start and aggressive driving, not during stabilized driving (Graham et al. 2008), and especially at cold test temperatures (Aakko-Saksa et al. 2011). Acetaldehyde emissions are efficiently removed by the catalyst, which reduces risk associated acetaldehyde emissions (CRFA 2003).
Ethanol emissions are high for ethanol containing fuels at cold-start, and especially at cold temperatures (Aakko-Saksa et al. 2011). However, with warmed-up engine ethanol emissions are typically below detection limit with E10 and E20 fuels (Graham et al. 2008). Ethanol may hung-up in vehicle system leading to carry-over effect (ethanol found even with E0 fuel).
Particulate matter, PAH and mutagenicity
Particulate matter emission (PM) is not considered as significant emission from conventional gasoline fuelled cars, whereas PM from direct-injection gasoline cars may be substantial. Generally, ethanol reduces PM emissions compared to gasoline. In the study from Australia (2008) the PM emissions were 35% lower for E10 fuel than for gasoline with model year 2006 or newer cars.
Indication of lower particulate PAH emissions and mutagenicity of particulates with ethanol when compared to MTBE containing reformulated gasoline has been reported (Pentikäinen 2004, Aakko 2003). Aakko-Saksa et al. (2011) reported that ethanol may reduce particulate matter associated PAHs from direct-injection car. The same trend was not seen for Ames mutagenicity. For gasoline cars with indirect injection technology, particulate matter emission levels were low and fuel related differences were not observed.
AFDC (2009) summarized that when tailpipe and evaporative emissions with E10 are considered, ozone forming potential of exhaust is increasing when compared to gasoline. This is due to increased evaporative emissions, acetaldehyde emissions and NOx emissions. However, Graham et al. (2008) reported that E10 and E20 blends did not affect the ozone forming potential for the MPFI gasoline cars, andt decreased ozone forming potential for a direct-injection gasoline car. Aakko-Saksa et al. (2011) did not observe significant effect of low-level ethanol blends on ozone forming potential.
Australian study (2008) modeled photochemical smog using Sydney as a sample area. In these conditions, an increase in peak ozone concentration was seen in E5/E10 scenarios. Reduction in tailpipe HC and CO emissions with ethanol did not compensate the increase in volatile organic emissions.
Car technology has developed drastically in 10-20 years. Many of the studies with low concentration ethanol blends have been carried out with cars that do not represent today’s technology. Emission level from new cars tends to be low, and thus the effect of fuel on the exhaust emissions is small at normal temperature. Differences in emissions between fuels are mainly seen at cold-start, heavy-driving conditions and at cold temperatures.
The major drawback of adding low-level ethanol into gasoline is an increase in volatile organic compounds and acetaldehyde emissions. Exhaust catalyst can efficiently remove aldehyde emissions, but it is not operating properly e.g. during cold-start. The same applies to ethanol emissions.
Ethanol has generally a positive impact on tailpipe CO and HC emissions. For NOx emissions, contradictory results are reported. Benzene and 1,3-butadiene emissions seem not to be influenced by ethanol content of gasoline, if olefins and aromatics contents of fuel do not change. Particulate matter emissions generally decrease with ethanol containing gasoline.
Ozone forming potential may increase with ethanol/gasoline blends due to the increased evaporative and acetaldehyde emissions. However, this phenomenon is complex process dependent on many factors, such as NOx emissions and climatic conditions.
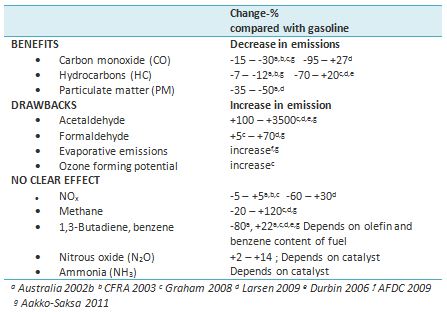
Summary of emission studies is shown in Figure 3 and Table 2.
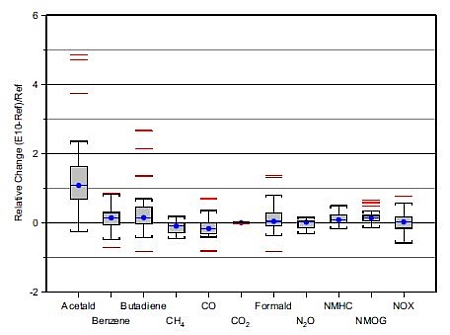
Figure 3. The effect of E10 on exhaust emissions (Graham 2008).
Table 2. Changes in emissions when E10 is compared to gasoline (negative values = reduction in emissions, positive values = increase in emissions).